Chapter 2
BCG Failure: Definitions & Management
Understanding the spectrum of BCG failure — from BCG-unresponsive to BCG-exposed disease — is critical for appropriate treatment selection and patient counselling.
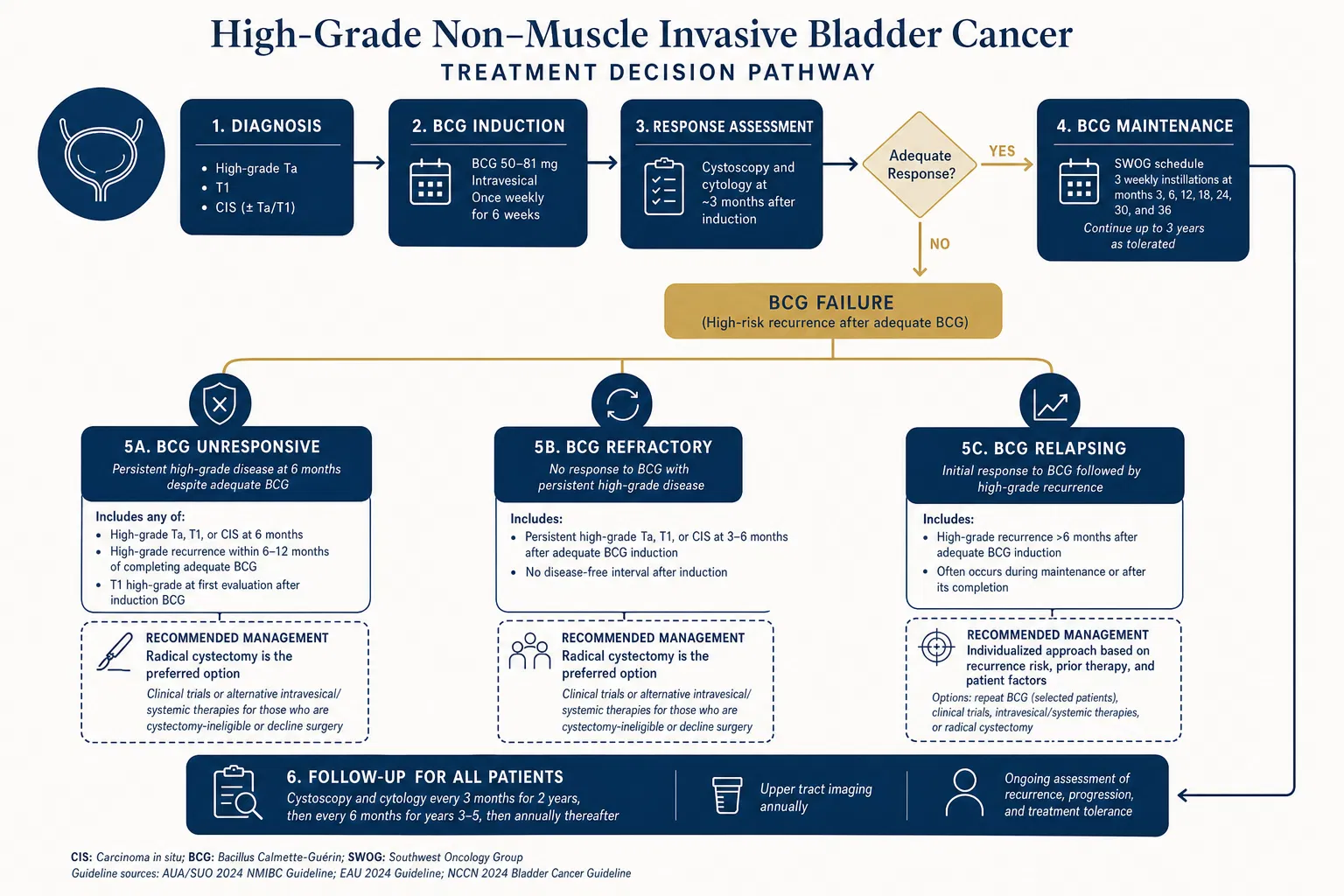
Figure 2. Treatment decision pathway for high-grade NMIBC — from diagnosis through BCG failure management.
The Spectrum of BCG Failure
Despite being the most effective intravesical therapy for high-risk NMIBC, BCG fails in approximately 30–50% of patients. The terminology surrounding BCG failure has evolved significantly, with the 2018 FDA guidance document and subsequent AUA/EAU guideline updates providing standardised definitions to facilitate clinical trial design and guide treatment decisions.
The concept of "adequate BCG" is fundamental to these definitions. Adequate BCG is defined as receipt of at least 5 of 6 doses during the initial induction course, plus either at least 2 of 3 doses of maintenance therapy or at least 2 of 6 doses of a second induction course. This distinction is crucial because patients who have not received adequate BCG may still benefit from further BCG therapy.
BCG-Unresponsive Disease
BCG-unresponsive NMIBC represents the most treatment-resistant category and is defined by the 2018 FDA guidance as any of the following scenarios after adequate BCG therapy:
Persistent/Recurrent CIS
Carcinoma in situ persisting or recurring within 12 months of completing adequate BCG therapy.
Recurrent High-Grade Ta/T1
High-grade papillary disease (Ta or T1) recurring within 6 months of completing adequate BCG therapy.
High-Grade T1 at First Evaluation
T1 high-grade disease detected at the first cystoscopic evaluation following a BCG induction course.
"Patients with BCG-unresponsive disease should be offered radical cystectomy. For those who are ineligible or unwilling, FDA-approved alternatives include pembrolizumab, nadofaragene firadenovec, and nogapendekin alfa inbakicept."
— AUA/SUO NMIBC Guideline, 2024BCG-Refractory vs. BCG-Relapsing
While the FDA definition focuses on "BCG-unresponsive" as a unified category, the EAU guidelines provide additional granularity by distinguishing between BCG-refractory and BCG-relapsing disease:
| Category | Definition | Prognosis |
|---|---|---|
| BCG-Refractory | Persistent high-grade disease at 6 months despite adequate BCG, or any stage/grade progression by 3 months after first BCG cycle | Worst prognosis |
| BCG-Relapsing | Recurrence of high-grade disease after achieving a disease-free state following adequate BCG; occurs >6 months after last BCG | Intermediate |
| BCG-Exposed | Recurrence after inadequate BCG, or high-risk recurrence 12–24 months after adequate BCG | May respond to further BCG |
| BCG-Naïve Recurrence | Any recurrence >24 months after completing adequate BCG induction + maintenance | Treat as new diagnosis |
Management Algorithm
The management of BCG failure depends critically on the specific category of failure, patient fitness for radical surgery, and patient preferences. The following algorithm reflects current AUA/EAU guideline recommendations:
Confirm BCG Failure Category
Verify adequate BCG exposure, timing of recurrence, and histological grade/stage. Perform re-staging TURBT and upper tract imaging.
BCG-Unresponsive: Offer Radical Cystectomy
Radical cystectomy with pelvic lymph node dissection remains the gold standard for BCG-unresponsive disease, offering the best long-term oncological outcomes.
Bladder-Sparing Options (if cystectomy-ineligible/unwilling)
FDA-approved alternatives: Pembrolizumab (systemic), Nadofaragene firadenovec (intravesical gene therapy), Nogapendekin alfa + BCG (intravesical). Off-label: Gemcitabine/Docetaxel, clinical trials.
BCG-Exposed/Relapsing: Consider Repeat BCG
Patients with BCG-exposed or late-relapsing disease may benefit from additional BCG therapy. Recent data suggest rescue BCG achieves 75% disease-free rates in selected patients.
Prognostic Factors in BCG Failure
Several factors influence outcomes following BCG failure and should guide treatment decision-making. The presence of T1 disease (vs. Ta or CIS alone), concurrent CIS with papillary disease, lymphovascular invasion, variant histology (micropapillary, plasmacytoid, nested), and early timing of recurrence (<6 months) all portend worse outcomes and should lower the threshold for recommending radical cystectomy.
Conversely, patients with isolated CIS recurrence, late relapse (>12 months), absence of variant histology, and good performance status may be reasonable candidates for bladder-sparing approaches, particularly with the availability of novel FDA-approved agents.
Key References
- FDA Guidance: BCG-Unresponsive Nonmuscle Invasive Bladder Cancer: Developing Drugs and Biologics for Treatment. 2018.
- Holzbeierlein JM, et al. AUA/SUO Guideline: Diagnosis and Treatment of NMIBC. 2024 Amendment. J Urol. 2024.
- Naselli A, et al. BCG Refractory NMIBC: Current Guidance and Experience from Clinical Practice. Res Rep Urol. 2024;16:299-305.
- EAU Guidelines on Non-muscle-invasive Bladder Cancer (TaT1 and CIS). 2024 Update.
- Myers A, et al. Challenging the Paradigm of "BCG Unresponsive" Bladder Cancer: Does Additional BCG Have an Effect? AUA 2024.