Chapter 3
Novel FDA-Approved Agents
Three breakthrough therapies have received FDA approval for BCG-unresponsive NMIBC between 2020 and 2024, offering bladder-sparing alternatives for patients who are ineligible or unwilling to undergo radical cystectomy.
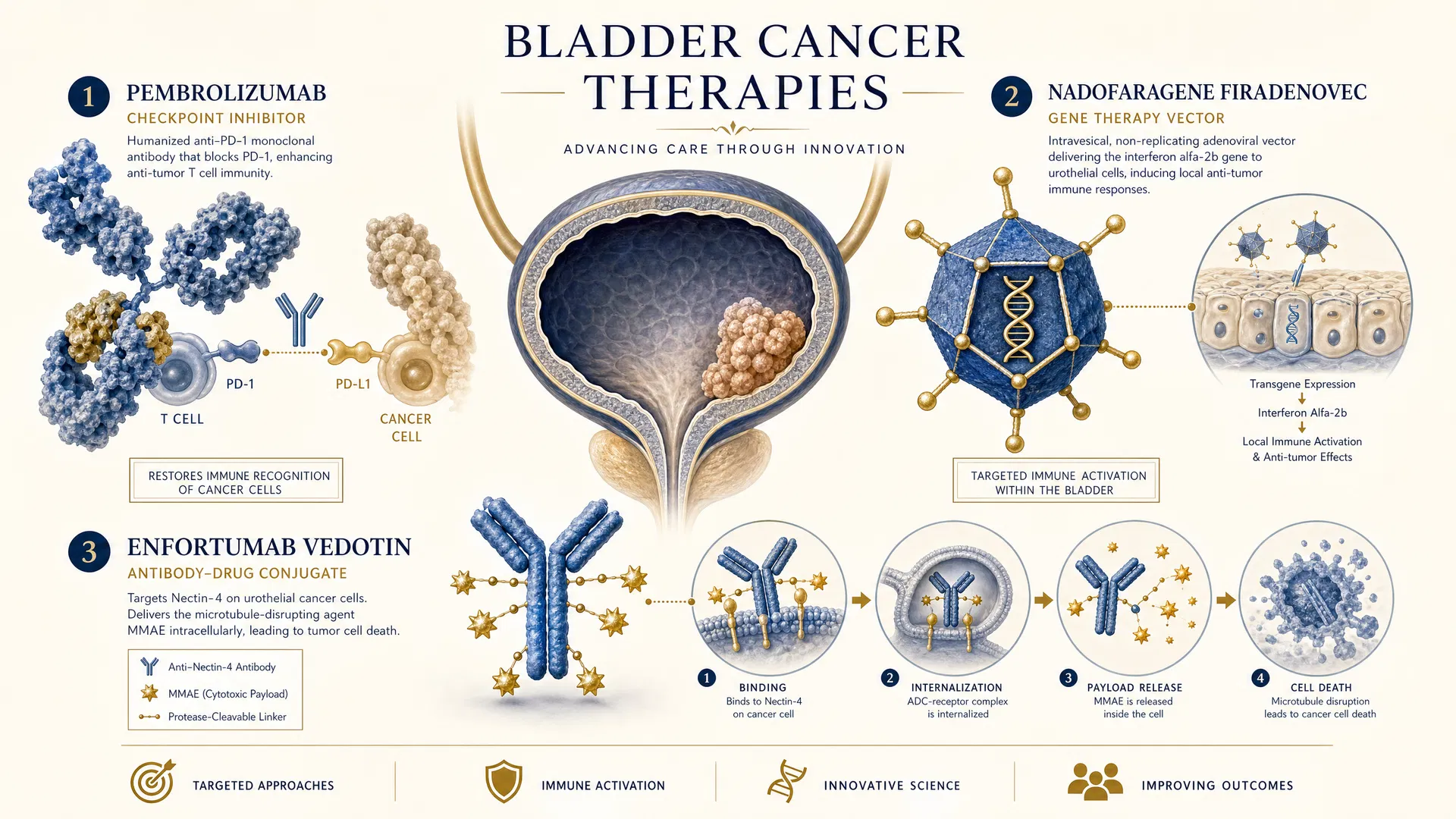
Figure 3. Novel therapeutic agents for BCG-unresponsive NMIBC — mechanisms of action.
Therapeutic Landscape Evolution
The period from 2020 to 2024 has witnessed a transformative shift in the management of BCG-unresponsive NMIBC. For decades, radical cystectomy was the only guideline-recommended option for these patients, with intravesical valrubicin (approved 1998) offering limited efficacy. The approval of three mechanistically distinct agents — a checkpoint inhibitor, a gene therapy vector, and an IL-15 receptor agonist — has fundamentally expanded the treatment armamentarium.
Pembrolizumab (Keytruda)
January 2020
Intravenous (systemic)
Anti-PD-1 checkpoint inhibitor
KEYNOTE-057
BCG-unresponsive, high-risk NMIBC with CIS (± Ta/T1)
Pembrolizumab is a humanised monoclonal antibody targeting the programmed death-1 (PD-1) receptor on T lymphocytes. By blocking the PD-1/PD-L1 interaction, pembrolizumab restores T-cell mediated anti-tumour immunity that is often suppressed in the tumour microenvironment. It was the first systemic immunotherapy approved for NMIBC.
The KEYNOTE-057 trial (Cohort A) enrolled 148 patients with BCG-unresponsive CIS with or without papillary disease. Patients received pembrolizumab 200 mg IV every 3 weeks for up to 24 months. The complete response (CR) rate was 41% at 3 months, with a median duration of response of 16.2 months. Approximately 46% of responders maintained CR at 12 months.
"Pembrolizumab represents the first systemic immunotherapy to demonstrate meaningful clinical activity in BCG-unresponsive NMIBC, with durable responses observed in a significant proportion of patients."
Treatment-related adverse events of grade 3–4 severity occurred in 13% of patients, consistent with the known safety profile of pembrolizumab in other indications. Immune-related adverse events included hypothyroidism (8%), colitis (2%), and pneumonitis (1%). No treatment-related deaths were reported.
Nadofaragene Firadenovec (Adstiladrin)
December 2022
Intravesical
Non-replicating adenoviral vector delivering IFN-α2b gene
Phase III (Boorjian et al.)
BCG-unresponsive, high-risk NMIBC (CIS, Ta, T1)
Nadofaragene firadenovec is a first-in-class, non-replicating adenoviral vector-based gene therapy that delivers the human interferon alfa-2b (IFN-α2b) gene directly to urothelial cells. Upon intravesical instillation, the adenoviral vector transduces bladder epithelial cells, which then produce and secrete IFN-α2b locally, creating sustained high concentrations of this anti-tumour cytokine within the bladder microenvironment.
The pivotal Phase III trial enrolled 157 patients with BCG-unresponsive high-risk NMIBC (103 with CIS ± Ta/T1, 54 with high-grade Ta/T1 without CIS). Nadofaragene was administered intravesically every 3 months. In the CIS cohort, the complete response rate was 53.4% at 3 months, with 24.3% maintaining CR at 12 months. In the papillary-only cohort, the high-grade recurrence-free rate was 72.9% at 12 months.
The safety profile was favourable, with the most common adverse events being bladder-related (dysuria 11%, urgency 7%, haematuria 6%). No grade 4 or 5 treatment-related adverse events were reported. The intravesical route of administration avoids systemic immune-related toxicities seen with checkpoint inhibitors.
Nogapendekin Alfa Inbakicept (Anktiva)
April 2024
Intravesical (combined with BCG)
IL-15 receptor agonist (IL-15 superagonist/IL-15Rα-Fc fusion)
QUILT-3.032
BCG-unresponsive NMIBC with CIS (± Ta/T1)
Nogapendekin alfa inbakicept is a first-in-class IL-15 receptor agonist that combines a novel IL-15 superagonist with the IL-15 receptor alpha-Fc fusion protein. This complex activates and expands NK cells and CD8+ T cells — key effectors of anti-tumour immunity — while avoiding the activation-induced cell death that limits native IL-15 signalling. Critically, it is administered in combination with BCG, potentially synergising with BCG's immunostimulatory effects.
The QUILT-3.032 trial enrolled 77 patients with BCG-unresponsive NMIBC with CIS. Patients received intravesical nogapendekin alfa combined with BCG. The complete response rate was 62% at any time point, with 52% maintaining CR at 12 months — the highest durable response rate reported among approved agents for this indication.
"Nogapendekin alfa inbakicept combined with BCG achieved the highest complete response rate and 12-month durability among FDA-approved therapies for BCG-unresponsive NMIBC with CIS."
The combination was generally well tolerated, with the most common adverse events being local bladder symptoms (dysuria, frequency, urgency) and flu-like symptoms consistent with immune activation. Grade 3 adverse events occurred in approximately 20% of patients, with no grade 4–5 treatment-related events.
Comparative Overview
| Agent | Route | CR Rate | 12-mo CR | Key AE |
|---|---|---|---|---|
| Pembrolizumab | IV q3w | 41% | ~19% | irAEs (thyroid, colitis) |
| Nadofaragene | Intravesical q3mo | 53.4% | 24.3% | Dysuria, urgency |
| Nogapendekin + BCG | Intravesical | 62% | 52% | Bladder symptoms, flu-like |
Note: Cross-trial comparisons should be interpreted with caution due to differences in patient populations, endpoints, and study designs.
Treatment Selection Considerations
The choice among these agents depends on multiple factors including disease characteristics (CIS vs. papillary), patient comorbidities, prior autoimmune conditions (which may preclude pembrolizumab), BCG availability (required for nogapendekin alfa), and patient preference regarding systemic vs. local therapy. Ongoing real-world studies and head-to-head comparisons will further refine treatment algorithms.
For patients with CIS-predominant disease, nogapendekin alfa + BCG currently offers the highest reported CR rate and durability. For patients with contraindications to BCG or those preferring to avoid systemic therapy, nadofaragene firadenovec provides an attractive intravesical-only option. Pembrolizumab may be preferred for patients with concurrent upper tract disease or those who may benefit from systemic immune activation.
Key References
- Balar AV, et al. Pembrolizumab monotherapy for the treatment of high-risk non-muscle-invasive bladder cancer unresponsive to BCG (KEYNOTE-057): an open-label, single-arm, multicentre, phase 2 study. Lancet Oncol. 2021;22(7):919-930.
- Boorjian SA, et al. Intravesical nadofaragene firadenovec gene therapy for BCG-unresponsive non-muscle-invasive bladder cancer: a single-arm, open-label, repeat-dose clinical trial. Lancet Oncol. 2021;22(1):107-117.
- Chamie K, et al. Intravesical nogapendekin alfa inbakicept-pmln plus BCG for BCG-unresponsive non-muscle-invasive bladder cancer (QUILT-3.032). J Clin Oncol. 2024.
- FDA Approval: Nogapendekin alfa inbakicept-pmln for BCG-unresponsive NMIBC. April 22, 2024.
- Tyson MD. Factors Influencing the Selection of Nadofaragene Firadenovec in BCG-Unresponsive NMIBC. OncLive. 2025.