Chapter 4
Gemcitabine/Docetaxel & Intravesical Enfortumab Vedotin
Sequential intravesical gemcitabine and docetaxel offers a well-tolerated alternative after BCG failure, while intravesical enfortumab vedotin represents an exciting frontier in targeted intravesical therapy.
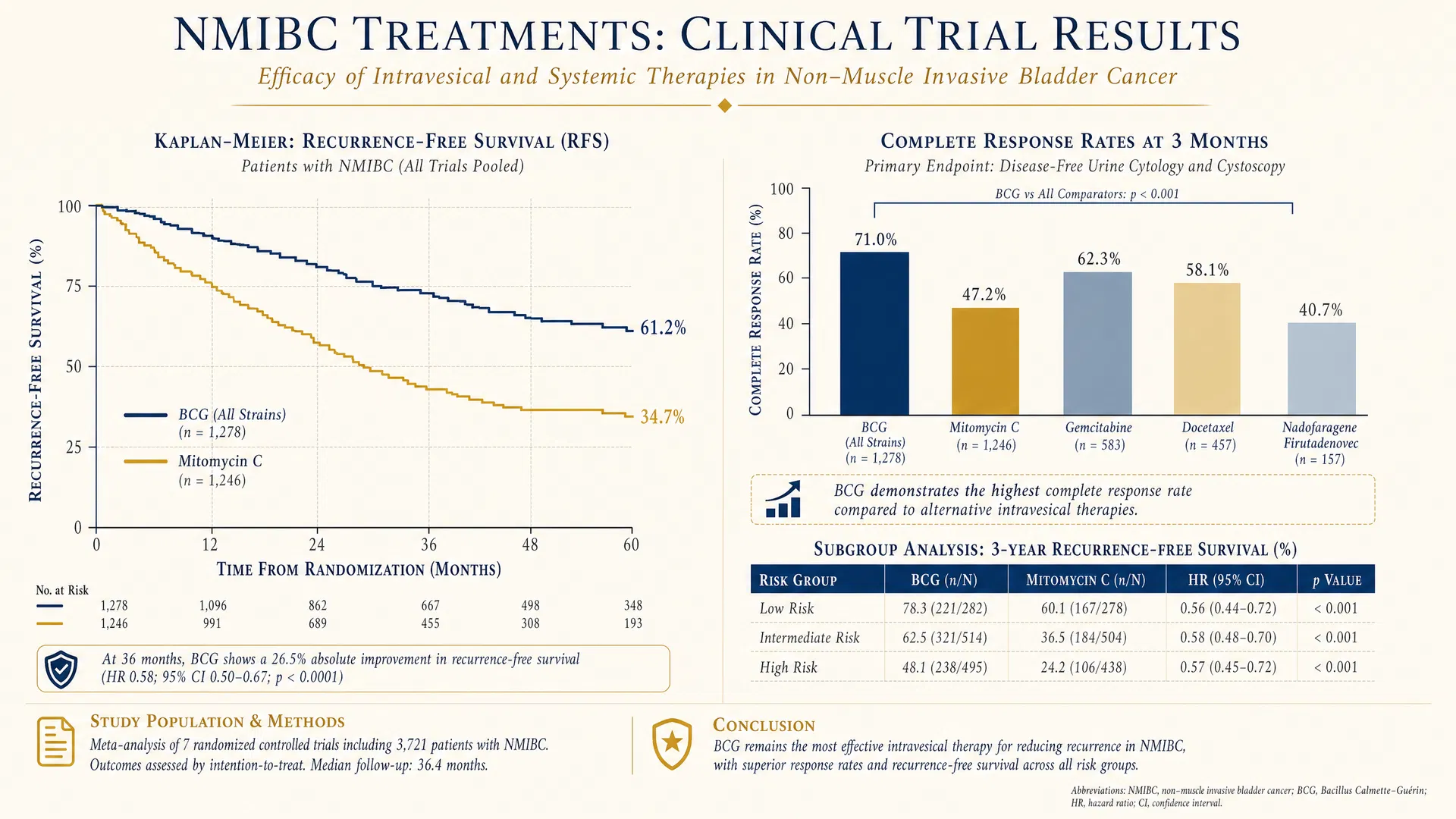
Figure 4. Clinical trial results — comparative efficacy of intravesical and systemic therapies in NMIBC.
Sequential Gemcitabine/Docetaxel (Gem/Doce)
Sequential intravesical gemcitabine and docetaxel has emerged as one of the most widely adopted off-label regimens for patients with BCG failure who are not candidates for or decline radical cystectomy. Originally described by Steinberg et al., this combination leverages the complementary mechanisms of a nucleoside analogue (gemcitabine) and a taxane (docetaxel) to achieve synergistic anti-tumour activity within the bladder.
The rationale for sequential administration is based on preclinical data demonstrating that gemcitabine sensitises tumour cells to subsequent taxane-mediated cytotoxicity by inhibiting DNA repair mechanisms. The intravesical route ensures high local drug concentrations while minimising systemic exposure and toxicity.
Treatment Protocol
The standard Gem/Doce protocol involves sequential intravesical administration during each treatment session:
Gem/Doce Instillation Protocol
Gemcitabine Instillation
1000 mg gemcitabine in 50 mL sterile saline. Dwell time: 60–90 minutes. Patient voids after dwell period.
Docetaxel Instillation
37.5 mg docetaxel in 50 mL sterile saline. Administered immediately after voiding gemcitabine. Dwell time: 60–120 minutes.
Induction Schedule
Weekly instillations for 6 consecutive weeks.
Maintenance Schedule
Monthly instillations for up to 24 months (if disease-free at 3-month assessment).
Clinical Evidence
The largest published series of intravesical Gem/Doce comes from a multi-institutional retrospective analysis of 276 patients with high-risk NMIBC after BCG failure (Steinberg et al., ASCO 2022). At a median follow-up of 22.5 months, the high-grade recurrence-free survival was 60% at 1 year and 46% at 2 years. The cystectomy-free survival was 85% at 2 years.
A subsequent analysis with longer follow-up (median 38 months) confirmed the durability of responses, with 3-year high-grade recurrence-free survival of 38% and cystectomy-free survival of 78%. These results compare favourably with historical data for other salvage intravesical regimens and support Gem/Doce as a reasonable bladder-sparing option.
"Sequential intravesical gemcitabine and docetaxel provides a durable response in patients with BCG failure, offering an effective and well-tolerated alternative to early cystectomy."
— Milbar et al., Bladder Cancer, 2023| Endpoint | 1 Year | 2 Years | 3 Years |
|---|---|---|---|
| HG Recurrence-Free Survival | 60% | 46% | 38% |
| Cystectomy-Free Survival | 92% | 85% | 78% |
| Progression-Free Survival | 95% | 88% | 82% |
Advantages of Gem/Doce
The Gem/Doce regimen offers several practical advantages over FDA-approved alternatives. Both agents are readily available, relatively inexpensive, and do not require cold-chain storage or specialised preparation facilities. The regimen is well tolerated, with grade 3–4 adverse events reported in fewer than 5% of patients. Common side effects are limited to mild dysuria, frequency, and occasional chemical cystitis, which are self-limiting.
Furthermore, the EAU Oncology Section has recently endorsed Gem/Doce as a viable option for European centres, noting its excellent results in retrospective analyses and its potential role as a bridge therapy while awaiting novel agent availability or clinical trial enrolment.
Intravesical Enfortumab Vedotin (EV)
Enfortumab vedotin (EV) is an antibody-drug conjugate (ADC) targeting Nectin-4, a cell adhesion molecule highly expressed on urothelial carcinoma cells. EV consists of a fully human anti-Nectin-4 monoclonal antibody conjugated to monomethyl auristatin E (MMAE), a potent microtubule-disrupting agent. Upon binding to Nectin-4 on tumour cells, the ADC is internalised and MMAE is released intracellularly, leading to mitotic arrest and cell death.
While EV is FDA-approved for intravenous use in locally advanced or metastatic urothelial carcinoma (based on the EV-301 trial demonstrating overall survival benefit), its intravesical application in NMIBC represents a novel and potentially transformative approach that leverages direct tumour exposure while minimising systemic toxicity.
EV-104: First-in-Human Intravesical EV Trial
Phase 1
Ongoing (interim results ASCO 2023)
BCG-unresponsive NMIBC
Dose-escalation, open-label
The EV-104 trial (NCT05014139) is a first-in-human, Phase 1 dose-escalation study evaluating the safety and preliminary efficacy of intravesical enfortumab vedotin in patients with NMIBC. Interim results presented at ASCO 2023 (Kamat et al.) reported data from 6 patients across 2 dose levels.
At the 125 mg dose level, 4 patients completed 6 induction cycles of weekly intravesical EV. The treatment was well tolerated with no dose-limiting toxicities observed. Preliminary anti-tumour activity was demonstrated, with evidence of tumour response on surveillance cystoscopy. Importantly, systemic exposure to MMAE was minimal, suggesting that intravesical administration achieves high local concentrations without significant systemic absorption.
"Intravesical enfortumab vedotin was well tolerated and showed antitumor activity in patients with NMIBC. These first clinical data support further investigation of intravesical EV as a novel treatment approach."
— Kamat AM et al., ASCO 2023 (Abstract 4596)The trial continues to enrol patients at higher dose levels, with expansion cohorts planned to further characterise efficacy. If confirmed in larger studies, intravesical EV could represent a paradigm shift in NMIBC treatment by combining the precision of targeted therapy with the favourable safety profile of local administration.
Future Directions
The intravesical therapy landscape for NMIBC continues to evolve rapidly. Several promising approaches are in clinical development, including intravesical TAR-200 (a gemcitabine-releasing intravesical device), CG0070 (an oncolytic adenovirus), vicinium/oportuzumab monatox (an EpCAM-targeted immunotoxin), and various combination strategies pairing intravesical agents with systemic checkpoint inhibitors.
The concept of "total intravesical therapy" — combining multiple local agents with complementary mechanisms — is gaining traction as a strategy to maximise anti-tumour efficacy while preserving the bladder. As more data mature from ongoing trials, treatment algorithms will likely become increasingly personalised based on molecular biomarkers, disease characteristics, and patient factors.
Key References
- Steinberg RL, et al. Long-term follow-up of intravesical gemcitabine and docetaxel for NMIBC after BCG failure. J Clin Oncol. 2022;40(suppl 6):573.
- Milbar N, et al. Sequential intravesical gemcitabine/docetaxel provides a durable response in BCG failure. Bladder Cancer. 2023;9(3):287-295.
- Kamat AM, et al. EV-104: A first-in-human trial of intravesical enfortumab vedotin in patients with NMIBC. J Clin Oncol. 2023;41(suppl 16):4596.
- EAU Oncology Section. Gemcitabine/Docetaxel: A New Option for European Centres in the Management of NMIBC. 2025.
- Wiesen B, et al. Updated review on novel therapies and ongoing clinical trials in NMIBC. Bladder Cancer. 2025.