Chapter 1
BCG Immunotherapy
Bacillus Calmette-Guérin remains the gold standard intravesical treatment for intermediate- and high-risk non-muscle invasive bladder cancer, with proven efficacy in reducing both recurrence and progression.
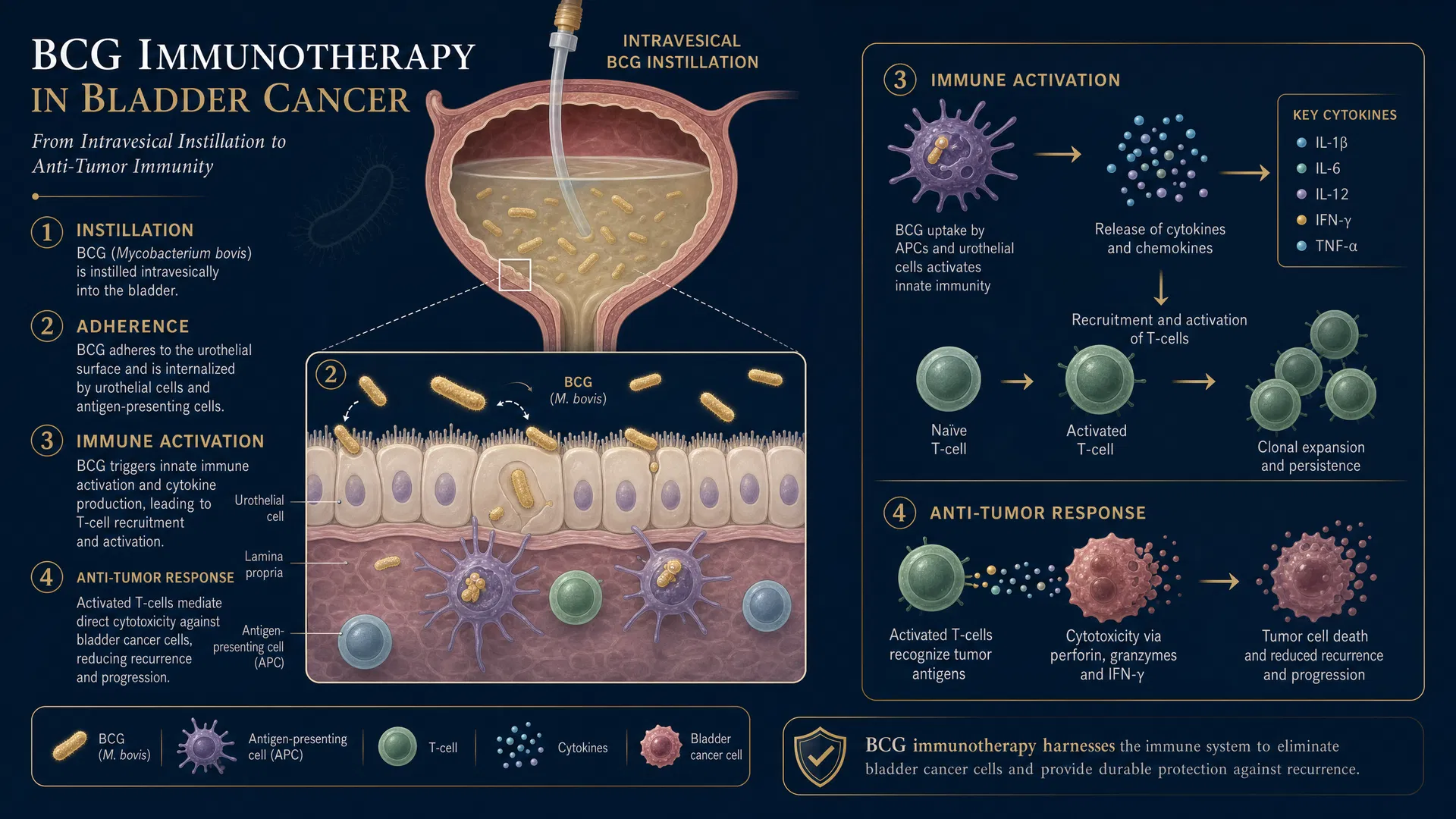
Figure 1. Mechanism of BCG immunotherapy — from intravesical instillation to anti-tumour immune response.
Historical Background
Bacillus Calmette-Guérin (BCG) is an attenuated strain of Mycobacterium bovis, originally developed as a vaccine against tuberculosis in 1921. Its application in bladder cancer was pioneered by Morales, Eidinger, and Bruce in 1976, who first demonstrated that intravesical BCG instillation could induce tumour regression. Since then, multiple randomised controlled trials and meta-analyses have confirmed BCG's superiority over intravesical chemotherapy in reducing recurrence and, critically, disease progression in high-risk NMIBC.
The landmark EORTC 30911 trial (Sylvester et al., 2010) and the subsequent Cochrane systematic review (Schmidt et al., 2020) established BCG with maintenance as the standard of care for high-risk NMIBC. BCG is the only intravesical agent that has consistently demonstrated a reduction in progression to muscle-invasive disease.
Mechanism of Action
BCG exerts its anti-tumour effect through a complex immunological cascade. Following intravesical instillation, BCG organisms attach to the urothelial surface via fibronectin and integrin receptors. The bacteria are then internalised by both urothelial cells and antigen-presenting cells (APCs), triggering innate immune activation.
This initial response leads to the release of pro-inflammatory cytokines including IL-1β, IL-6, IL-12, IFN-γ, and TNF-α, which recruit and activate CD4+ and CD8+ T lymphocytes, natural killer (NK) cells, and macrophages. The resulting Th1-dominant immune response mediates direct cytotoxicity against tumour cells through perforin/granzyme pathways and Fas/FasL interactions.
"BCG immunotherapy harnesses the immune system to eliminate bladder cancer cells and provide durable protection against recurrence through sustained T-cell mediated anti-tumour immunity."
Treatment Protocol
Induction Phase
BCG induction consists of six weekly intravesical instillations, typically initiated 2–4 weeks after TURBT to allow adequate wound healing. Each instillation involves reconstituting lyophilised BCG (available strains include TICE, Connaught/OncoTICE, and Tokyo-172) in 50 mL of preservative-free saline, administered via urethral catheter with a recommended dwell time of 1–2 hours.
The standard dose varies by strain: TICE BCG contains approximately 1–8 × 10⁸ colony-forming units (CFU), while the Tokyo strain uses 80 mg per instillation. Patients should be instructed to retain the solution for the full dwell period and to void in a seated position, with household bleach added to the toilet for 15 minutes before flushing.
Maintenance Phase (SWOG Protocol)
The Southwest Oncology Group (SWOG) maintenance protocol, established by Lamm et al. (2000), consists of three weekly instillations administered at months 3, 6, 12, 18, 24, 30, and 36 following the initial induction course. This 3-year maintenance schedule has demonstrated superior recurrence-free survival compared to induction alone.
SWOG Maintenance Schedule
Note: For high-risk NMIBC, full 3-year maintenance is recommended. For intermediate-risk, 1-year maintenance may suffice.
Clinical Evidence & Outcomes
Meta-analyses consistently demonstrate BCG's superiority over intravesical chemotherapy. BCG with maintenance reduces recurrence by approximately 32% and progression by 27% compared to mitomycin C. The 5-year recurrence-free survival for high-risk NMIBC patients receiving BCG with full maintenance ranges from 50–70%, depending on initial risk factors.
| Outcome | BCG + Maintenance | BCG Induction Only | Mitomycin C |
|---|---|---|---|
| 5-yr Recurrence-Free | 60–70% | 40–50% | 35–45% |
| 5-yr Progression-Free | 80–90% | 70–80% | 65–75% |
| Complete Response (CIS) | 70–75% | 55–65% | 40–50% |
Adverse Effects & Contraindications
BCG is generally well tolerated, though local and systemic side effects are common. Local symptoms include dysuria (60–80%), frequency (60–70%), and haematuria (40–50%), which typically resolve within 48 hours. Low-grade fever occurs in 20–30% of patients. Serious complications such as BCG sepsis (<1%), granulomatous prostatitis, and epididymitis are rare but require prompt treatment with anti-tuberculous therapy.
Absolute contraindications include active tuberculosis, immunosuppression, gross haematuria (risk of systemic absorption), traumatic catheterisation, and within 2 weeks of TURBT or bladder biopsy. Relative contraindications include urinary tract infection (should be treated prior to instillation) and prior severe BCG reaction.
Global BCG Shortage
Since 2012, the global supply of intravesical BCG has been intermittently compromised due to manufacturing difficulties and limited producers. This shortage has necessitated dose-reduction strategies (one-third dose) and prioritisation protocols, with guidelines recommending that available BCG be reserved for high-risk patients requiring induction therapy. The shortage has also accelerated research into alternative intravesical agents and combination regimens.
Key References
- Morales A, Eidinger D, Bruce AW. Intracavitary Bacillus Calmette-Guérin in the treatment of superficial bladder tumors. J Urol. 1976;116(2):180-183.
- Lamm DL, et al. Maintenance bacillus Calmette-Guérin immunotherapy for recurrent TA, T1 and carcinoma in situ transitional cell carcinoma of the bladder: a randomized Southwest Oncology Group Study. J Urol. 2000;163(4):1124-1129.
- Sylvester RJ, et al. Long-term efficacy results of EORTC genito-urinary group randomized phase 3 study 30911. Eur Urol. 2010;57(5):766-773.
- Schmidt S, et al. Intravesical Bacillus Calmette-Guérin versus mitomycin C for Ta and T1 bladder cancer. Cochrane Database Syst Rev. 2020;1(1):CD011935.
- Holzbeierlein JM, et al. Diagnosis and Treatment of Non-Muscle Invasive Bladder Cancer: AUA/SUO Guideline 2024 Amendment. J Urol. 2024;211(4):533-538.